DuoFertility
| DuoFertility | |
|---|---|
|
|
| Category | Torso-mounted |
| Developer | Cambridge Temperature Concepts Ltd [1] |
| Announced | 2005 [2] |
| Released | Developers:
Consumers: 2012 [3] |
| Price | 440 USD (May 2016, price for the 3 month plan)[4] |
| Operating system | none |
| Sensors |
temperature [5] |
| Weight | g (negligible) |
| Controls |
buttons, smartphone, desktop |
| Data available | Robust |
| Risk factor | Low |
| Not Standalone | |
| https://www.duofertility.com | |
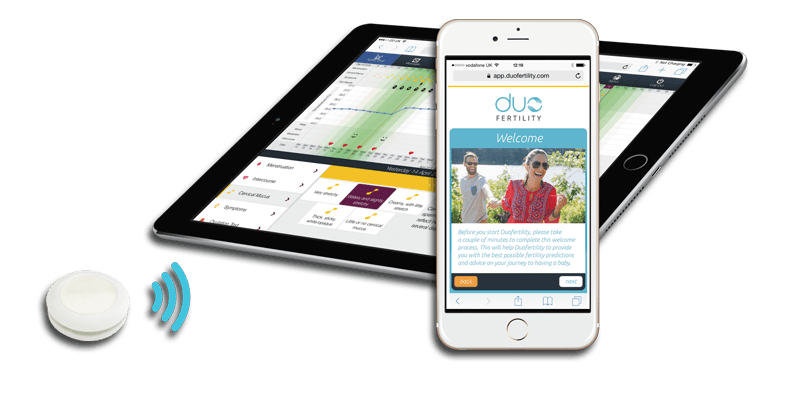
DuoFertility is a wearable temperature sensor that measures the changes in body temperature which occur during ovulation. The goal of the manufacturer is to help women with conception since they will be now able to monitor when they are the most fertile. The company claims a success rate of conception that is comparable to in vitro fertilisation.
Contents
Main Characteristics
The device consists of two separate pieces, the body temperature monitor, and the processing unit that wirelessly connects to the provided mobile app.
DuoFertility temperature sensors constantly monitors the changes in body temperature and sends the data to be analysed and stored in the processing unit. The user can also log various aspects of their menstruation cycle. All the gathered data are analysed and can be viewed in the provided mobile app. The user can make the decision to conceive based on the data however the company also provides support from fertility specialists as a part of the purchase. These are able to access and interpret the data and can offer advice on when will be the best time to conceive.
Purpose
The purpose of the device is to help pair with infertility by analysing the data about the women's ovulation and offering advice on when is the best time to conceive.
Company & People
DuoFertility is developed by a British company Cambridge Temperature Concepts Ltd. The company is based in Cambridge and was established in 2005. It focuses on the development of continuous physiological monitoring devices.[6][7]
- Dr. Andy Richards - Chairman of board of directors and advisors
- Dr Matthew Frohn - Board of directors and advisors member
- Pietrojan Gilardini - Board of directors and advisors member
- Dr Claire Hooper - Chief executive officer[8]
Important Dates
- 2005 - Cambridge Temperature Concepts founded.[2]
- January 2009 - The device obtains European Medical Devices Directive approval.[2]
- December 2011 - DuoFertility receives FDA clearance for the US market.[9]
- April 2012 - Commercial launch for the United States.[3]
Enhancement/Therapy/Treatment
DuoFertility helps couples to better predict the timespan, when the women is the most fertile, and thus enhances their ability to conceive.[10]
Ethical & Health Issues
We found no ethical or health issues that are relevant to this device in particular. However, some issues connected to wearables in general may still apply.
Public & Media Impact and Presentation
There are numerous articles about DuoFertility to be found, as well as user reviews and discussions.
Mobihealthnews has several articles in which DuoFertility is mentioned.[11] The first one discusses the presentation of DuoFertility at the Wireless Life-Sciences Alliance in San Diego in 2011. The article stated that the device was among the surprising presentation during that event and that it "really does span the continuum of health care" thanks to its incorporation of wireless technology.[12] Another article informs about the device receiving the approval of the US Food and Drug Administration and that it took almost two years to receive.[13] Next article informs about DuoFertility receiving the FDA approval in the US. It explains what the device is and what is does in general terms and concludes with an official press release by Cambridge Temperature Concepts Ltd.[14] Next article discusses wearables targeted on women and mentions DuoFertility among other products centred on female ovulation.[15] The similar approach is used in another article that compares the device to other fertility trackers.[16] And the latest article devoted to DuoFertility informs about the company securing more funds in order to "to expand product sales in the US and develop the next iteration of the DuoFertility tracker, which will offer smartphone connectivity and the ability to measure more metrics".[17]
TechCrunch magazine article opens with a then CEO of the company Shamus Husheer stating that, in comparison to other similar devices, DuoFertility has a much higher success rate. The article continues with a description on the device's features with Husheer's commentary included. The article concludes with the history of the company and the story of how they entered the American market.[18]
The Telegraph discusses DuoFertility at the time of publishing the research that shows the device is comparable to IVF fertility treatment. It glosses over the device's features and concludes with stating that the technology has a bog potential of helping pairs trying to conceive.[19]
The Guardian featured an expansive article describing the technological approach to tracking fertility by various companies, with the concluding part of the article focusing on DuoFertility. The article goes into detail on how the device can and also cannot help its user.[20]
In addition to the reviews and announcements, we registered a number of user reviews and comments about the device.
A blog especially devoted to DuoFertility describes the device as easy to use. The author of the blog was trying to conceive unsuccessfully and selected DuoFertility as an option cheaper to IVF. In the span of several months, the blog describes the author's struggle to get pregnant whilst using several devices including DuoFertility. She described the interface of the DuoFertility software as "great" and "accurate" and the temperatures sensor as "comfortable to wear". In comparison to other similar device the author used, she found DuoFertility to be much more accurate. After 8 months of trying the author successfully conceived which she credited to DuoFertility.[21]
4 Amazon users gave DuoFertility an average of 2.7 stars out of five. From which two reviews gave 5 stars, one 2 stars, and one 1 star. The positive reviews had positive experience with the device and especially with the included consulting programme. Both users attributed their pregnancy to DuoFertility. The negative reviews had bad experience with the consultants and said the device was complicated to use despite the high price. Both negative reviews concluded with saying that would not recommend the device to anyone.[22]
Mumsnet internet forum features a discussion on DuoFertility. The user are curious about the efficacy of the device and service and in general, the response from other users familiar with the device was negative. The overall sentiment was that the device can only recommend a good time to conceive and nothing else which, together with the steep price, makes it a difficult choice. Some users mentioned that DuoFertility is comparable to other devices and methods, since it was statistically proven that couples that have well timed intercourse can achieve pregnancy within 12 months anyway.[23]
Public Policy
DuoFertility has been approved by the Food and Drug Administration (FDA) For the US market.[24] DuoFertility has been approved by the Medicines and Healthcare products Regulatory Agency.[25]
Related Technologies, Projects, or Scientific Research
In a pilot study with 8 infertile women, DuoFertility was able to identify ovulation with 100% accuracy within one day.[26] A cohort study with 200 couples revealed that the success rate of conception assisted by DuoFertility is statistically comparable to in vitro fertilisation.[27]
There are various study, which focus on DuoFertility. They were conducted, however, by the employees of Cambridge Temperature Concepts Ltd. or in the cooperation with this company.
- CHAUSIAUX, O. E., et al. Are BMI, age, FSH and AMH good markers of spontaneous conception in infertile women?. Giornale Italiano di Ostetricia e Ginecologia. 2013, 35(1), 169-173.
- CHAUSIAUX, O. E., et al. BETTER THAN IVF? PREGNANCY RATES OF THE DUOFERTILITY PROGRAMME OVER SIX CYCLES OF USE. Reproductive BioMedicine Online (Reproductive Healthcare Limited). 2010. 20, S53-S54. DOI: 10.1016/S1472-6483(10)62544-8
- CHAUSIAUX, O. E., et al. The role of expectant management in treatment of couples with unexplained infertility. Giornale Italiano di Ostetricia e Ginecologia. 2013, 35(1), 174-179.
- ROLLASON, Jennie C. B., OUTTRIM, Joanne G., MATHUR Raj S. A pilot study comparing the DuoFertility® monitor with ultrasound in infertile women. International Journal of Women's Health. 2014, 6, 657–662. DOI: 10.2147/IJWH.S59080.
References
- ↑ https://www.duofertility.com/about-us/contact/
- ↑ 2.0 2.1 2.2 http://www.businesswire.com/news/home/20090107005556/en/Cambridge-Temperature-Concepts-gains-medical-approval-revolutionary
- ↑ 3.0 3.1 http://mobihealthnews.com/17150/duofertility-commercially-launches-in-us-for-795
- ↑ https://www.duofertility.com/buy/
- ↑ https://www.duofertility.com/what-is-duofertility/your-guide-to-using-duofertility/
- ↑ http://www.temperatureconcepts.com/
- ↑ https://www.crunchbase.com/organization/cambridge-temperature-concepts#/entity
- ↑ http://www.temperatureconcepts.com/about-us/
- ↑ http://mobihealthnews.com/16028/duofertility-wireless-sensor-receives-fda-clearance/
- ↑ https://www.duofertility.com/clinically-proven/
- ↑ http://mobihealthnews.com/tag/duofertility
- ↑ http://mobihealthnews.com/11042/wireless-health-opportunity-begins-at-conception
- ↑ http://mobihealthnews.com/16028/duofertility-wireless-sensor-receives-fda-clearance
- ↑ http://mobihealthnews.com/17150/duofertility-commercially-launches-in-us-for-795
- ↑ http://mobihealthnews.com/19907/digital-health-fertility-services-ramping-up
- ↑ http://mobihealthnews.com/30449/five-digital-health-tools-for-tracking-fertility
- ↑ http://mobihealthnews.com/35450/duofertility-maker-raises-4m-to-improve-fertility-tracker-add-smartphone-connection
- ↑ http://techcrunch.com/2013/04/01/duofertility/
- ↑ http://www.telegraph.co.uk/news/health/news/8069642/British-fertility-device-as-effective-as-IVF.html
- ↑ https://www.theguardian.com/technology/2014/may/18/desperate-pregnancy-app-smartphone-technology-couples-infertility
- ↑ https://duofertilityandclearblue.wordpress.com/
- ↑ https://www.amazon.com/DuoFertility-Fertility-Premium-Monitor-795/dp/B008OHGGB8#customerReviews
- ↑ http://www.mumsnet.com/Talk/conception/1547447-Duofertility-Anyone-tried-it
- ↑ http://www.accessdata.fda.gov/cdrh_docs/pdf10/k102499.pdf
- ↑ http://www.temperatureconcepts.com/digital-health-connecting-clinicians-and-patients-better-outcomes/
- ↑ http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4106957/
- ↑ http://www.sciencedirect.com/science/article/pii/S1472648310625448

